ILSI Europe disseminates science by publishing articles on original research, literature reviews and gap analyses, and meeting proceedings in peer-reviewed journals with high impact factors. ILSI Europe also publishes books, monographs, white papers, and other reports.
Discover below our latest scientific publications.
Publication Date
All Publications
Infant fat mass and later child and adolescent health outcomes: a systematic review
Archives of Disease in Childhood, 2023
- ILSI Europe
Obesity and excess adiposity are leading causes of metabolic and cardiovascular morbidity and mortality. Early identification of individuals at risk is key for preventive strategies. We examined the relationship between infant body composition (0–2 years of age) and later (>2 years) health outcomes using a systematic review.
Small intestine vs. colon ecology and physiology: Why it matters in probiotic administration
Cell Reports Medicine, 2023
- ILSI Europe
We provide a detailed review unfolding how the physiological and anatomical differences between the small and large intestine affect gut microbiota composition, function, and plasticity. This information is key to understanding how gut microbiota manipulation, including probiotic administration, may strain-dependently transform host-microbe interactions at defined locations.
An Overview of Approaches for Analysing NIAS from different FCMs
ILSI Europe Report Series , 2023
- ILSI Europe
This overview gives an excellent introduction to analysing NIAS. It considers non-harmonised FCMs, highlighting various FCMs and their unique requirements for analysis of migrants (IAS (Intentionally Added Substances) or NIAS). It presents background information on different test conditions for the different FCMs and why the standard tests used for plastics outlined in the Commission Regulation (EU) No 10/2011 (14 January 2011 on plastic materials and articles intended to come into contact with food) are inappropriate in some instances for many non-harmonised FCMs.
Impacts of the Ukraine–Russia Conflict on the Global Food Supply Chain and Building Future Resilience
EuroChoices, 2023
- ILSI Europe
The immediate concerns and potential solutions to the global impacts of the Ukraine–Russia conflict were recently discussed by a panel of industry, academic and civil society experts convened by the European branch of the International Life Sciences Institute, all of whom are authors of this manuscript.
Health relevance of lowering postprandial glycaemia in the paediatric population through diet’: results from a multistakeholder workshop
European Journal of Nutrition , 2022
- ILSI Europe
To summarize current knowledge and gaps regarding the role of postprandial glycaemic response in the paediatric population, a workshop was organized in June 2021 by the European branch of the International Life Science Institute (ILSI). The workshop led to the consensus on the crucial role on health of postprandial glycaemic response in paediatric population.
WP_Query Object
(
[query] => Array
(
[post_type] => publication
[posts_per_page] => 5
[type] =>
[area] =>
[committee] =>
[authors] =>
[showtitle] =>
[meta_query] => Array
(
[relation] => AND
)
[tax_query] => Array
(
)
[paged] => 1
[meta_key] => _ilsi_date
[orderby] => meta_value
[order] => DESC
)
[query_vars] => Array
(
[post_type] => publication
[posts_per_page] => 5
[type] =>
[area] =>
[committee] =>
[authors] =>
[showtitle] =>
[meta_query] => Array
(
[relation] => AND
)
[tax_query] => Array
(
)
[paged] => 1
[meta_key] => _ilsi_date
[orderby] => meta_value
[order] => DESC
[error] =>
[m] =>
[p] => 0
[post_parent] =>
[subpost] =>
[subpost_id] =>
[attachment] =>
[attachment_id] => 0
[name] =>
[pagename] =>
[page_id] => 0
[second] =>
[minute] =>
[hour] =>
[day] => 0
[monthnum] => 0
[year] => 0
[w] => 0
[category_name] =>
[tag] =>
[cat] =>
[tag_id] =>
[author] =>
[author_name] =>
[feed] =>
[tb] =>
[meta_value] =>
[preview] =>
[s] =>
[sentence] =>
[title] =>
[fields] =>
[menu_order] =>
=>
[category__in] => Array
(
)
[category__not_in] => Array
(
)
[category__and] => Array
(
)
[post__in] => Array
(
)
[post__not_in] => Array
(
)
[post_name__in] => Array
(
)
[tag__in] => Array
(
)
[tag__not_in] => Array
(
)
[tag__and] => Array
(
)
[tag_slug__in] => Array
(
)
[tag_slug__and] => Array
(
)
[post_parent__in] => Array
(
)
[post_parent__not_in] => Array
(
)
[author__in] => Array
(
)
[author__not_in] => Array
(
)
[search_columns] => Array
(
)
[ignore_sticky_posts] =>
[suppress_filters] =>
[cache_results] => 1
[update_post_term_cache] => 1
[update_menu_item_cache] =>
[lazy_load_term_meta] => 1
[update_post_meta_cache] => 1
[nopaging] =>
[comments_per_page] => 50
[no_found_rows] =>
)
[tax_query] => WP_Tax_Query Object
(
[queries] => Array
(
)
[relation] => AND
[table_aliases:protected] => Array
(
)
[queried_terms] => Array
(
)
[primary_table] => wp_3_posts
[primary_id_column] => ID
)
[meta_query] => WP_Meta_Query Object
(
[queries] => Array
(
[0] => Array
(
[key] => _ilsi_date
)
[relation] => OR
)
[relation] => AND
[meta_table] => wp_3_postmeta
[meta_id_column] => post_id
[primary_table] => wp_3_posts
[primary_id_column] => ID
[table_aliases:protected] => Array
(
[0] => wp_3_postmeta
)
[clauses:protected] => Array
(
[wp_3_postmeta] => Array
(
[key] => _ilsi_date
[compare] => =
[compare_key] => =
[alias] => wp_3_postmeta
[cast] => CHAR
)
)
[has_or_relation:protected] =>
)
[date_query] =>
[request] =>
SELECT SQL_CALC_FOUND_ROWS wp_3_posts.ID
FROM wp_3_posts INNER JOIN wp_3_postmeta ON ( wp_3_posts.ID = wp_3_postmeta.post_id )
WHERE 1=1 AND (
wp_3_postmeta.meta_key = '_ilsi_date'
) AND ((wp_3_posts.post_type = 'publication' AND (wp_3_posts.post_status = 'publish' OR wp_3_posts.post_status = 'acf-disabled')))
GROUP BY wp_3_posts.ID
ORDER BY wp_3_postmeta.meta_value DESC
LIMIT 0, 5
[posts] => Array
(
[0] => WP_Post Object
(
[ID] => 13987
[post_author] => 351
[post_date] => 2023-11-09 08:52:48
[post_date_gmt] => 2023-11-09 08:52:48
[post_content] =>
Objective
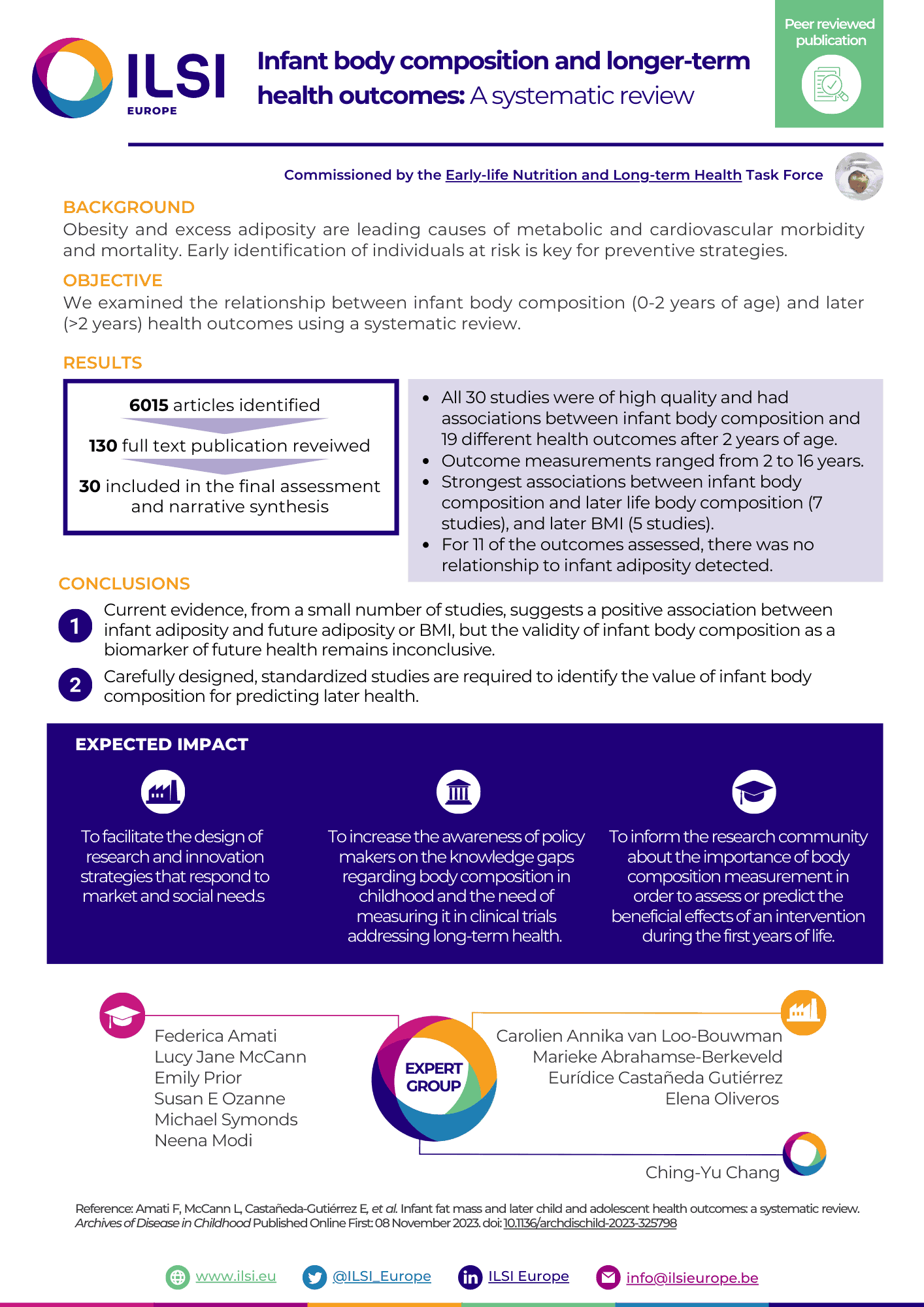
Obesity and excess adiposity are leading causes of metabolic and cardiovascular morbidity and mortality. Early identification of individuals at risk is key for preventive strategies. We examined the relationship between infant body composition (0-2 years of age) and later (>2 years) health outcomes using a systematic review.
Design
We preregistered the study on PROSPERO (ID 288013) and searched Embase, PubMed and Cochrane databases for English language publications using the Medical Subject Headings (MeSH) terms 'infant' and 'body composition' and 'risk' between January 1946 and February 2022. We included studies which assessed infant body composition using predetermined in vivo methods other than body mass index (BMI).
Results
We identified 6015 articles. After abstract screening to assess eligibility, we reviewed 130 full text publications. 30 were included in the final assessment and narrative synthesis. Meta-analysis was not possible due to heterogeneity of results. All 30 studies were of high quality and reported associations between infant body composition and 19 different health outcomes after 2 years of age. Outcome measurements ranged from 2 years to 16 years. The strongest associations were found between infant fat mass and later fat mass (7 studies), and later BMI (5 studies). For 11 of the outcomes assessed, there was no relationship to infant adiposity detected.
Conclusions
Current evidence, from a small number of studies, suggests a positive association between infant adiposity and future adiposity or BMI, but the validity of infant body composition as a biomarker of future health remains inconclusive. Carefully designed, standardised studies are required to identify the value of infant body composition for predicting later health.
Download the full article here.
Download the article one-pager below
 [post_title] => Infant fat mass and later child and adolescent health outcomes: a systematic review
[post_excerpt] =>
[post_status] => publish
[comment_status] => closed
[ping_status] => closed
[post_password] =>
[post_name] => infant-fat-mass-and-later-child-and-adolescent-health-outcomes-a-systematic-review
[to_ping] =>
[pinged] =>
[post_modified] => 2024-03-15 15:13:16
[post_modified_gmt] => 2024-03-15 15:13:16
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://ilsi.eu/?post_type=publication&p=13987
[menu_order] => 0
[post_type] => publication
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[1] => WP_Post Object
(
[ID] => 13549
[post_author] => 351
[post_date] => 2023-09-08 08:55:39
[post_date_gmt] => 2023-09-08 08:55:39
[post_content] =>
[post_title] => Infant fat mass and later child and adolescent health outcomes: a systematic review
[post_excerpt] =>
[post_status] => publish
[comment_status] => closed
[ping_status] => closed
[post_password] =>
[post_name] => infant-fat-mass-and-later-child-and-adolescent-health-outcomes-a-systematic-review
[to_ping] =>
[pinged] =>
[post_modified] => 2024-03-15 15:13:16
[post_modified_gmt] => 2024-03-15 15:13:16
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://ilsi.eu/?post_type=publication&p=13987
[menu_order] => 0
[post_type] => publication
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[1] => WP_Post Object
(
[ID] => 13549
[post_author] => 351
[post_date] => 2023-09-08 08:55:39
[post_date_gmt] => 2023-09-08 08:55:39
[post_content] =>
Research on gut microbiota has generally focused on fecal samples, representing luminal content of the large intestine. However, nutrient uptake is restricted to the small intestine. Abundant immune cell populations at this anatomical site combined with diminished mucus secretion and looser junctions (partly to allow for more efficient fluid and nutrient absorption) also results in intimate host-microbe interactions despite more rapid transit. It is thus crucial to dissect key differences in both ecology and physiology between small and large intestine to better leverage the immense potential of human gut microbiota imprinting, including probiotic engraftment at biological sensible niches. Here, we provide a detailed review unfolding how the physiological and anatomical differences between the small and large intestine affect gut microbiota composition, function, and plasticity. This information is key to understanding how gut microbiota manipulation, including probiotic administration, may strain-dependently transform host-microbe interactions at defined locations.
This review focusses its narrative on the intimate relationship between the host and its bacterial constituents of the small and large intestine.
Dowload the full paper
or click on the images below to download the EG one-pager summary and graphical abstract

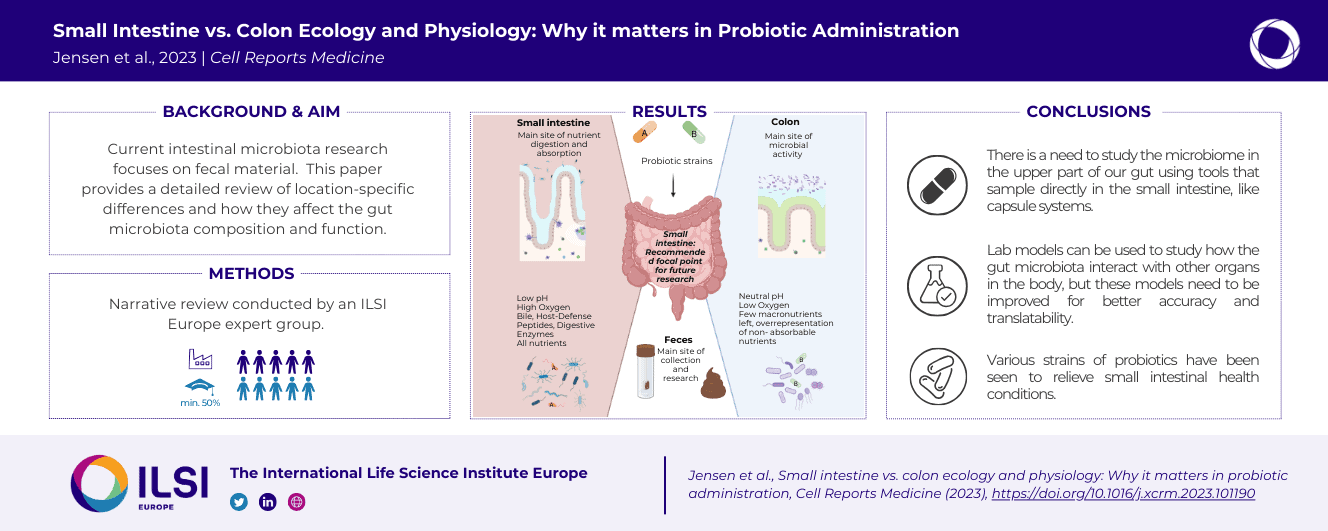
Commissioned by the Probiotics Task Force
[post_title] => Small intestine vs. colon ecology and physiology: Why it matters in probiotic administration
[post_excerpt] =>
[post_status] => publish
[comment_status] => closed
[ping_status] => closed
[post_password] =>
[post_name] => small-intestine-vs-colon-ecology-and-physiology-why-it-matters-in-probiotic-administration
[to_ping] =>
[pinged] =>
[post_modified] => 2023-09-08 12:36:10
[post_modified_gmt] => 2023-09-08 12:36:10
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://ilsi.eu/?post_type=publication&p=13549
[menu_order] => 0
[post_type] => publication
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[2] => WP_Post Object
(
[ID] => 12598
[post_author] => 351
[post_date] => 2023-04-06 09:57:58
[post_date_gmt] => 2023-04-06 09:57:58
[post_content] =>
[post_title] => An Overview of Approaches for Analysing NIAS from different FCMs
[post_excerpt] =>
[post_status] => publish
[comment_status] => closed
[ping_status] => closed
[post_password] =>
[post_name] => an-overview-of-approaches-for-analysing-nias-from-different-fcms
[to_ping] =>
[pinged] =>
[post_modified] => 2023-10-23 13:54:47
[post_modified_gmt] => 2023-10-23 13:54:47
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://ilsi.eu/?post_type=publication&p=12598
[menu_order] => 0
[post_type] => publication
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[3] => WP_Post Object
(
[ID] => 12507
[post_author] => 351
[post_date] => 2023-02-22 08:55:39
[post_date_gmt] => 2023-02-22 08:55:39
[post_content] =>
[post_title] => Impacts of the Ukraine–Russia Conflict on the Global Food Supply Chain and Building Future Resilience
[post_excerpt] =>
[post_status] => publish
[comment_status] => closed
[ping_status] => closed
[post_password] =>
[post_name] => impacts-of-the-ukraine-russia-conflict-on-the-global-food-supply-chain-and-building-future-resilience
[to_ping] =>
[pinged] =>
[post_modified] => 2023-06-19 12:44:01
[post_modified_gmt] => 2023-06-19 12:44:01
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://ilsi.eu/?post_type=publication&p=12507
[menu_order] => 0
[post_type] => publication
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[4] => WP_Post Object
(
[ID] => 12345
[post_author] => 24
[post_date] => 2023-01-04 12:00:18
[post_date_gmt] => 2023-01-04 12:00:18
[post_content] =>
[post_title] => Health relevance of lowering postprandial glycaemia in the paediatric population through diet’: results from a multistakeholder workshop
[post_excerpt] =>
[post_status] => publish
[comment_status] => closed
[ping_status] => closed
[post_password] =>
[post_name] => health-relevance-of-lowering-postprandial-glycaemia-in-the-paediatric-population-through-diet-results-from-a-multistakeholder-workshop
[to_ping] =>
[pinged] =>
[post_modified] => 2023-06-19 12:46:23
[post_modified_gmt] => 2023-06-19 12:46:23
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://ilsi.eu/?post_type=publication&p=12345
[menu_order] => 0
[post_type] => publication
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
)
[post_count] => 5
[current_post] => -1
[before_loop] =>
[in_the_loop] =>
[post] => WP_Post Object
(
[ID] => 13987
[post_author] => 351
[post_date] => 2023-11-09 08:52:48
[post_date_gmt] => 2023-11-09 08:52:48
[post_content] =>
Objective
Obesity and excess adiposity are leading causes of metabolic and cardiovascular morbidity and mortality. Early identification of individuals at risk is key for preventive strategies. We examined the relationship between infant body composition (0-2 years of age) and later (>2 years) health outcomes using a systematic review.
Design
We preregistered the study on PROSPERO (ID 288013) and searched Embase, PubMed and Cochrane databases for English language publications using the Medical Subject Headings (MeSH) terms 'infant' and 'body composition' and 'risk' between January 1946 and February 2022. We included studies which assessed infant body composition using predetermined in vivo methods other than body mass index (BMI).
Results
We identified 6015 articles. After abstract screening to assess eligibility, we reviewed 130 full text publications. 30 were included in the final assessment and narrative synthesis. Meta-analysis was not possible due to heterogeneity of results. All 30 studies were of high quality and reported associations between infant body composition and 19 different health outcomes after 2 years of age. Outcome measurements ranged from 2 years to 16 years. The strongest associations were found between infant fat mass and later fat mass (7 studies), and later BMI (5 studies). For 11 of the outcomes assessed, there was no relationship to infant adiposity detected.
Conclusions
Current evidence, from a small number of studies, suggests a positive association between infant adiposity and future adiposity or BMI, but the validity of infant body composition as a biomarker of future health remains inconclusive. Carefully designed, standardised studies are required to identify the value of infant body composition for predicting later health.
Download the full article here.
Download the article one-pager below
 [post_title] => Infant fat mass and later child and adolescent health outcomes: a systematic review
[post_excerpt] =>
[post_status] => publish
[comment_status] => closed
[ping_status] => closed
[post_password] =>
[post_name] => infant-fat-mass-and-later-child-and-adolescent-health-outcomes-a-systematic-review
[to_ping] =>
[pinged] =>
[post_modified] => 2024-03-15 15:13:16
[post_modified_gmt] => 2024-03-15 15:13:16
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://ilsi.eu/?post_type=publication&p=13987
[menu_order] => 0
[post_type] => publication
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[comment_count] => 0
[current_comment] => -1
[found_posts] => 429
[max_num_pages] => 86
[max_num_comment_pages] => 0
[is_single] =>
[is_preview] =>
[is_page] =>
[is_archive] =>
[is_date] =>
[is_year] =>
[is_month] =>
[is_day] =>
[is_time] =>
[is_author] =>
[is_category] =>
[is_tag] =>
[is_tax] =>
[is_search] =>
[is_feed] =>
[is_comment_feed] =>
[is_trackback] =>
[is_home] => 1
[is_privacy_policy] =>
[is_404] =>
[is_embed] =>
[is_paged] =>
[is_admin] =>
[is_attachment] =>
[is_singular] =>
[is_robots] =>
[is_favicon] =>
[is_posts_page] =>
[is_post_type_archive] =>
[query_vars_hash:WP_Query:private] => 66bebe078556f954d2e43ee5198dd4a9
[query_vars_changed:WP_Query:private] =>
[thumbnails_cached] =>
[allow_query_attachment_by_filename:protected] =>
[stopwords:WP_Query:private] =>
[compat_fields:WP_Query:private] => Array
(
[0] => query_vars_hash
[1] => query_vars_changed
)
[compat_methods:WP_Query:private] => Array
(
[0] => init_query_flags
[1] => parse_tax_query
)
)
[post_title] => Infant fat mass and later child and adolescent health outcomes: a systematic review
[post_excerpt] =>
[post_status] => publish
[comment_status] => closed
[ping_status] => closed
[post_password] =>
[post_name] => infant-fat-mass-and-later-child-and-adolescent-health-outcomes-a-systematic-review
[to_ping] =>
[pinged] =>
[post_modified] => 2024-03-15 15:13:16
[post_modified_gmt] => 2024-03-15 15:13:16
[post_content_filtered] =>
[post_parent] => 0
[guid] => https://ilsi.eu/?post_type=publication&p=13987
[menu_order] => 0
[post_type] => publication
[post_mime_type] =>
[comment_count] => 0
[filter] => raw
)
[comment_count] => 0
[current_comment] => -1
[found_posts] => 429
[max_num_pages] => 86
[max_num_comment_pages] => 0
[is_single] =>
[is_preview] =>
[is_page] =>
[is_archive] =>
[is_date] =>
[is_year] =>
[is_month] =>
[is_day] =>
[is_time] =>
[is_author] =>
[is_category] =>
[is_tag] =>
[is_tax] =>
[is_search] =>
[is_feed] =>
[is_comment_feed] =>
[is_trackback] =>
[is_home] => 1
[is_privacy_policy] =>
[is_404] =>
[is_embed] =>
[is_paged] =>
[is_admin] =>
[is_attachment] =>
[is_singular] =>
[is_robots] =>
[is_favicon] =>
[is_posts_page] =>
[is_post_type_archive] =>
[query_vars_hash:WP_Query:private] => 66bebe078556f954d2e43ee5198dd4a9
[query_vars_changed:WP_Query:private] =>
[thumbnails_cached] =>
[allow_query_attachment_by_filename:protected] =>
[stopwords:WP_Query:private] =>
[compat_fields:WP_Query:private] => Array
(
[0] => query_vars_hash
[1] => query_vars_changed
)
[compat_methods:WP_Query:private] => Array
(
[0] => init_query_flags
[1] => parse_tax_query
)
)
